Quantitative Chemical Analysis 10th Edition PDF is a comprehensive and authoritative guide to the fundamental principles, techniques, and applications of quantitative chemical analysis. This classic textbook has been meticulously crafted to provide a clear and accessible introduction to the subject, making it an indispensable resource for students, researchers, and professionals alike.
Delving into the intricacies of quantitative chemical analysis, this seminal work elucidates the concepts of accuracy, precision, and error, laying the foundation for understanding the nuances of analytical measurements. It meticulously examines the principles and methodologies of gravimetric, volumetric, and spectroscopic techniques, empowering readers with a deep comprehension of the diverse analytical approaches employed in modern chemistry.
Introduction to Quantitative Chemical Analysis
Quantitative chemical analysis is the science of determining the concentration or amount of a substance in a sample.
Quantitative chemical analysis is important in a variety of fields, including:
- Environmental monitoring
- Food analysis
- Pharmaceutical analysis
- Forensic science
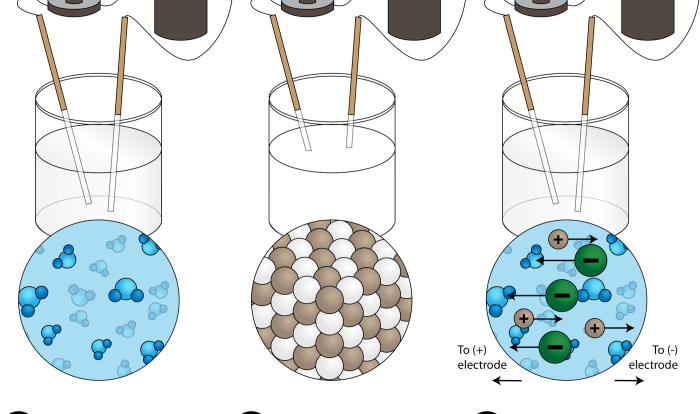
The principles of quantitative chemical analysis are based on the laws of chemistry. These laws allow us to determine the amount of a substance in a sample by measuring its physical or chemical properties.
Methods and Procedures in Quantitative Chemical Analysis
There are a variety of methods used in quantitative chemical analysis. The most common methods include:
- Gravimetric analysis
- Volumetric analysis
- Spectroscopic analysis
Each of these methods has its own advantages and disadvantages. The choice of method depends on the specific analyte being determined and the desired level of accuracy.
Data Analysis and Interpretation
The data from a quantitative chemical analysis must be analyzed and interpreted in order to be useful. The data can be used to:
- Determine the concentration of a substance in a sample
- Identify the presence of a substance in a sample
- Determine the purity of a substance
The data can also be used to develop calibration curves, which can be used to determine the concentration of a substance in future samples.
Applications of Quantitative Chemical Analysis, Quantitative chemical analysis 10th edition pdf
Quantitative chemical analysis is used in a variety of applications, including:
- Environmental monitoring
- Food analysis
- Pharmaceutical analysis
- Forensic science
- Quality control
- Product development
Quantitative chemical analysis is an essential tool for a variety of industries and professions.
Emerging Trends in Quantitative Chemical Analysis
There are a number of emerging trends in quantitative chemical analysis. These trends include:
- The use of advanced instrumentation
- The development of new analytical techniques
- The use of computational methods
These trends are helping to improve the accuracy, precision, and speed of quantitative chemical analysis.
FAQ Summary: Quantitative Chemical Analysis 10th Edition Pdf
What is the significance of quantitative chemical analysis?
Quantitative chemical analysis plays a pivotal role in various scientific fields, including environmental monitoring, food chemistry, and pharmaceutical development. It enables researchers and professionals to determine the precise composition and concentration of substances, providing crucial information for decision-making and quality control.
What are the key principles of quantitative chemical analysis?
Quantitative chemical analysis is guided by the principles of accuracy, precision, and error. Accuracy refers to the closeness of a measurement to the true value, while precision indicates the reproducibility of a measurement. Error analysis helps quantify and minimize uncertainties associated with measurements.
What are the different methods used in quantitative chemical analysis?
Quantitative chemical analysis employs a range of methods, including gravimetric analysis, volumetric analysis, and spectroscopic analysis. Gravimetric analysis involves determining the mass of a substance, while volumetric analysis measures the volume of a solution required to react with a substance.
Spectroscopic analysis utilizes the interaction of electromagnetic radiation with matter to identify and quantify substances.